- Hotline: 852+ 2891 9689
- Whatsapp: 852+ 9146 8426
- [email protected]
- 6/F 29 Austin Road, Tsimshatsui, Kowloon, Hong Kong
LLLT for Hair Loss & Post-Transplant Care: An Evidence-Based Adjunct Therapy
Home » Low Level Laser Therapy
When used in the immediate post-operative period, LLLT can be a beneficial adjunct. Its primary role is to reduce inflammation and edema, potentially creating a better environment for graft healing. However, it is not a substitute for surgical precision and does not correct poor technique. Any long-term growth benefits are secondary and require ongoing, consistent use.
 Sara’s Comment:
Sara’s Comment:LLLT is an FDA-cleared, non-invasive modality that can support hair follicle health. In our practice, its primary and most evidence-supported use is during the immediate post-transplant healing phase to reduce inflammation and support graft survival. For long-term hair growth stimulation, effects are modest and require consistent, long-term home use with a medical-grade device, making it a proven medical treatments.
LLLT, or photobiomodulation, aims to stimulate follicles in the anagen phase and awaken those stalled in telogen/kenogen. Its cellular effects, crucial for treatment timing, include:
Stimulating Mitochondrial Activity: Light energy is absorbed to enhance ATP (cellular energy) production in follicle cells.
Increasing Blood Flow: Promotes microcirculation, delivering more nutrients and oxygen to the follicle.
Reducing Inflammation: Modulates inflammatory markers that can inhibit growth.
Extending Anagen Phase: Shifts follicles from resting phases back into the active growth phase.
Foundation for Treatment: All effective hair loss interventions—from medication to surgery—are based on manipulating the hair growth cycle.
Phase-Specific Actions: Treatments work by either prolonging anagen (growth), shortening telogen (rest), or reactivating kenogen (latent) follicles.
Timing is Critical: Misapplied force or trauma during certain phases can worsen loss, as seen in poorly timed transplants or aggressive procedures.
LLLT Requires Precision: As a science-based therapy, LLLT’s success depends on correct wavelength, optimal dosing, and consistent application to safely stimulate follicles.
Primary Benefit: May reduce post-operative swelling, redness, and inflammation.
Potential Benefit: Could help minimize shock loss (telogen effluvium) and support early graft stabilization.
FDA-Cleared Treatment: It is a recognized modality for pattern hair loss.
Modest Efficacy: Studies show it can help slow thinning and improve hair density in some users, but results are typically less pronounced than with pharmaceutical treatments (minoxidil/finasteride).
Critical Requirement: Requires consistent, long-term use (typically every other day) with a properly calibrated device.
More than 1,000 publications have reported that laser or low energy lasers can effectively increase cell survival, proliferation and function. Clinical controlled trials have shown that lasers can stimulate and preserve hair follicles affected by androgenetic alopecia and other alopecia.
Primary Benefit is Post-Op Healing: LLLT’s best-supported use is reducing inflammation and supporting graft survival in the first week after a hair transplant.
Modest Long-Term Effects: For treating ongoing hair loss, LLLT can provide modest improvement in hair density but requires diligent, long-term use and is not a replacement for proven medications.
Device Quality is Critical: Effective treatment requires an FDA-cleared, medical-grade laser device; consumer LEDs lack proven efficacy.
Understand the Dose Response: More light is not better; incorrect dosing can be counterproductive. Protocols must be followed precisely.
An Adjunct, Not a Miracle: LLLT is a supportive tool within a broader treatment plan, not a standalone solution for significant hair restoration.
A professional assessment can determine if LLLT is a suitable adjunct for your case. Send your hair loss photos and questions for a preliminary review. We will provide an honest evaluation of whether LLLT—and in what form—could be a beneficial component of your medical or post-surgical regimen.

Images & Information shown are for reference only
Information on this website is provided for general educational purposes only and does not constitute personalized medical advice. It is not intended to promote our service or imply superiority over another.
Individual results in hair restoration vary significantly and no outcome can be guaranteed. The before-and-after images shown represent possible results — not promises. We recommend seeking independent medical advice to discuss your options … Read More
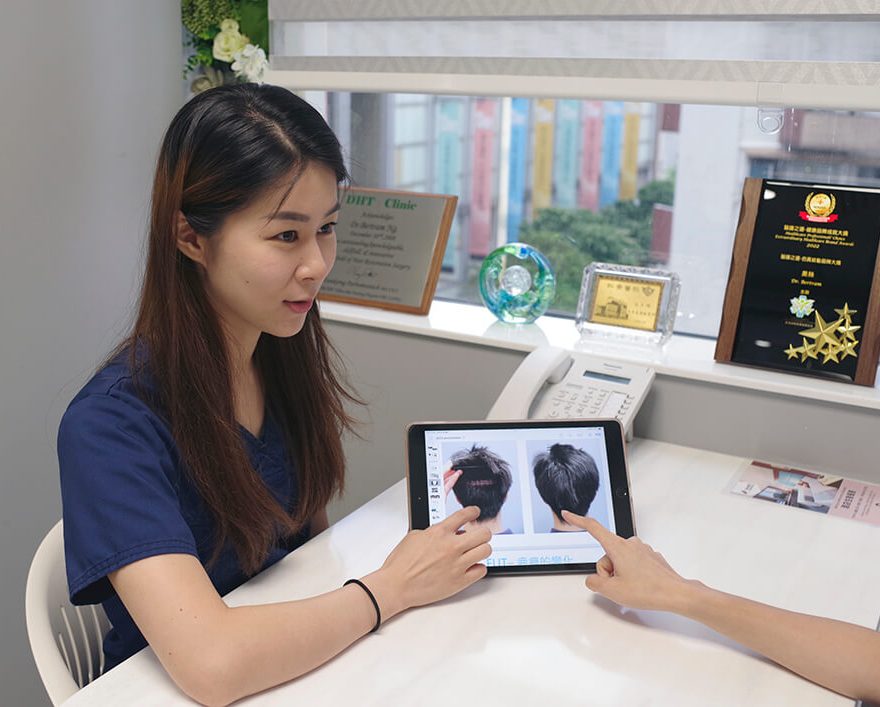
Pre-Consultation Review
If you want to get a personalized answer from our medical team, you can reach us using this form here. We will cantact you as soon as possible.
Our practice adheres to guidelines established by leading international organizations in Hair Restoration.

International Society of Hair Restoration Surgery is the leading global medical association that establishes international practice standards and patient safety protocols.

The American Board of Hair restoration Surgery represents the highest standard. To maintain rigorous certification requirements the physician must demonstrate surgical expertise.

Worls FUE InstituteI serves as the premier educational body focused exclusively on Follicular Unit Extraction methodology. The institute ensures consistent application of safe FUE.